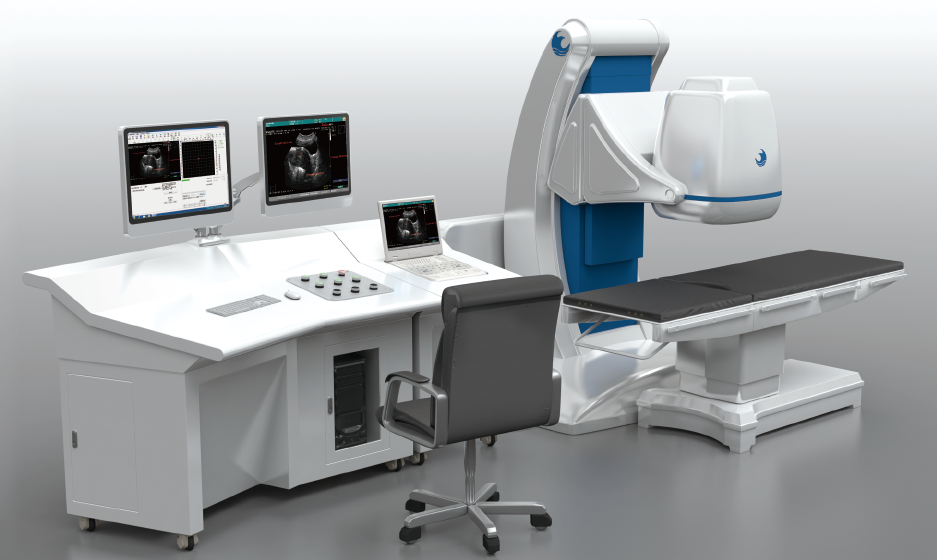
HY2900 HIFU
for Uterine Fibroids
Non-invasive focused ultrasound system for organ-preserving treatment of uterine fibroids and adenomyosis
*Regulated and approved by China’s National Medical Products Administration (NMPA, formerly CFDA) as a Class III high‑intensity focused ultrasound device. Included in China’s National Medical Insurance (NHSA) reimbursement catalogue.
About Liansheng Medtech
Advanced Focused Ultrasound for Women's Health
Liansheng Medtech is the exclusive global distributor of the HY2900 HIFU system, bringing cutting-edge non-invasive ultrasound technology to clinical partners across the region. We work directly with healthcare institutions to provide equipment, clinical support, and training for the treatment of uterine fibroids and adenomyosis.
5000+ cases
NMPA-approved Class III Medical Device
>90% efficacy Rate
<10% recurrence Rate
ISO 13485 Certified
<2 hours single Treatment
FDA/CE Pending Certification anticipated 2027
Covered under China’s National Medical Insurance (NHSA) Reimbursement catalogue for uterine fibroid treatment.
Endorsed by Ping An Insurance (China)- full reimbursement in the event of an ineffective treatment outcome.
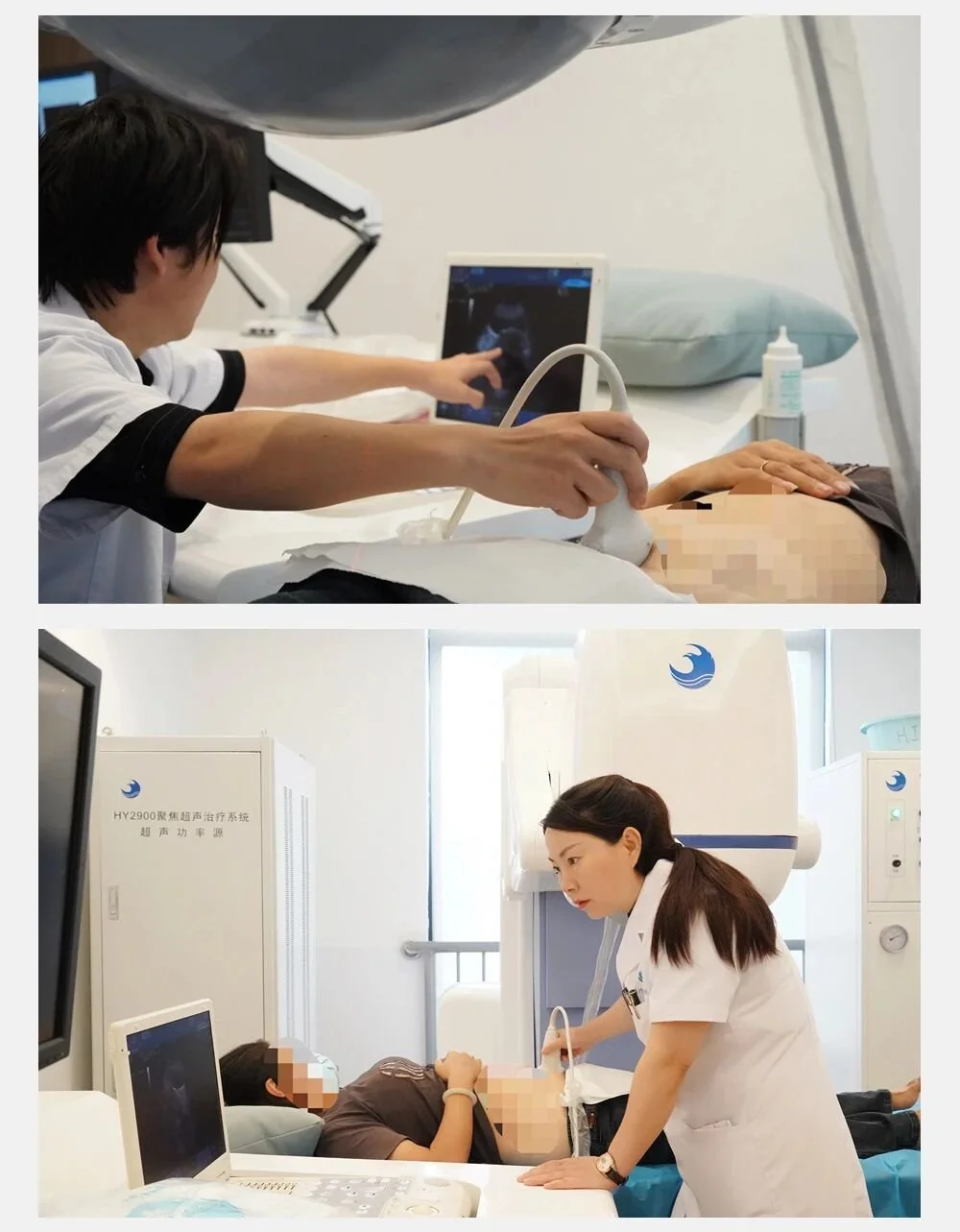



Focused Ultrasound Therapy (HIFU)
Focused ultrasound therapy (HIFU) is performed under medical imaging guidance, concentrating ultrasound waves on an internal target region to form a high–energy‑density focal point. The focal tissue temperature rises rapidly, producing coagulative necrosis within a very short time, while tissue outside the focus shows no obvious damage. By controlling the three‑dimensional movement of the focal point, conformal treatment of the entire target tissue is achieved.
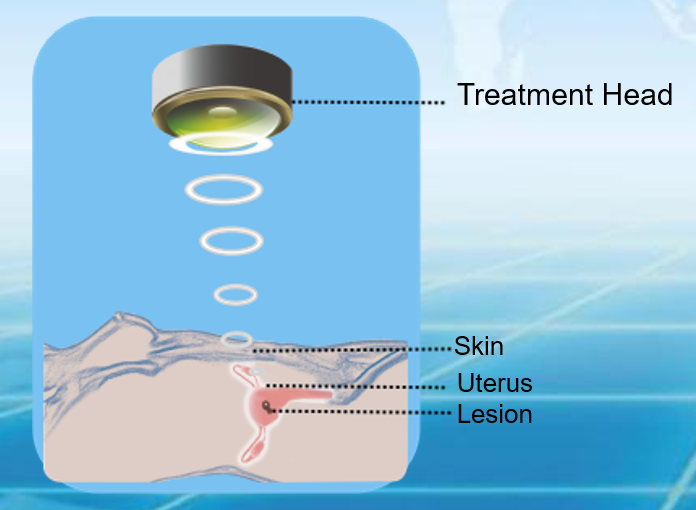
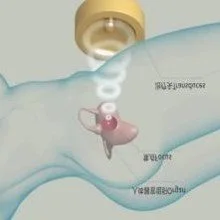
Focused ultrasound (HIFU) technology has been explored clinically in indications such as uterine fibroids, adenomyosis, benign prostatic hyperplasia, breast nodules, endometriosis, and scar pregnancy.
With a clinical record of 5,000+ successful treatments, the HY2900 system is specifically designed and clinically configured for non-invasive, uterus‑preserving treatment of uterinefibroids and adenomyosis.
Why choose the HY2900 HIFU SYSTEM
Treat. Heal. Preserve. The future of non-invasive tumor therapy.
KEY BENEFITS
Truly Non-invasive
No surgical incision needed. Ultrasound penetrates the body surface to directly ablate target tissue, avoiding the risks of traditional surgery.

Effective
Ablation with coagulative necrosis was achieved in 100% of treated lesions. At 6 months, 73% of tumors show ≥50% volume reduction; supplementary sessions deliver ≥95% reduction in remaining cases.
Safe
Over 5,000 procedures performed on the HY2900 system with no reported medical incidents. Patients can start planning pregnancy six months after treatment.
Rapid Recovery
Patients can typically walk independently within hours, with no hospitalization required. Most return to work within 1-2 days.
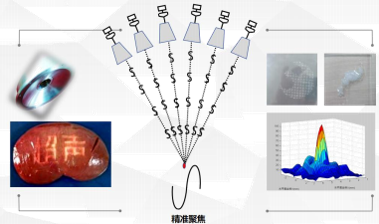
Precise
Focal spot accuracy: 1.2 × 1.2 × 8 mm³. 5D mechanical scanning accuracy: 0.1 mm. Phased‑array technology. Crown‑shaped treatment mode (proprietary technology).
Patient-friendly
Treatment performed by a single physician/ gynecologist without general anesthesia. Patient can communicate with physician during treatment.
Let’s Keep In Touch

Send us your details and we’ll follow up within 2 business days.
Find us at : jiahao.zhao@lianshengmed.com